9514. o-Tolidine
Nomenclature
CAS number: 119-93-7
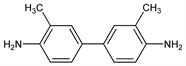
3,3′-Dimethyl-[1,1′-biphenyl]-4,4′-diamine; 3,3′-dimethylbenzidine; 4,4′-diamino-3,3′-dimethylbiphenyl. C14H16N2; mol wt 212.29.
C 79.21%, H 7.60%, N 13.20%.
Description and references
Made by alkaline reduction of o-nitrotoluene with zinc, and subsequent rearrangement of the o-hydrazotoluene formed, by boiling with HCl: Van Loon, Chem. Weekbl. 5, 689 (1907). See also Schultz et al., Ann. 352, 111 (1907). Crystal and molecular structure: Chawdhury et al., Acta Crystallogr. B24, 1222 (1968). Metabolism: Dieteren, Arch. Environ. Health 12, 30 (1966). Carcinogenic activity: Pliss, Zebenzhinskii, J. Natl. Cancer Inst. 45, 283 (1970).
Properties
White to reddish crystals or cryst powder. mp 129-131°. Slightly sol in water; sol in alcohol, ether, dil acids. Keep well closed and protected from light.Derivative
Sulfate.Nomenclature
CAS number: 531-20-4
C14H16N2.H2SO4; mol wt 310.37.
C 54.18%, H 5.85%, N 9.03%, S 10.33%, O 20.62%.
Properties
White to gray mass. Slightly sol in water, alcohol; sol in dil acids.Derivative
Dihydrochloride.Nomenclature
CAS number: 612-82-8
C14H16N2.2HCl; mol wt 285.21.
C 58.96%, H 6.36%, N 9.82%, Cl 24.86%.