9545. Tomatine
Nomenclature
Description and references
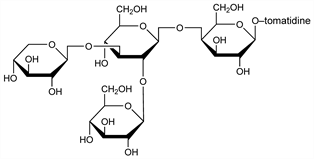
Occurs in the extract of leaves of wild tomato plants: Fontaine et al., Arch. Biochem. 18, 467 (1948); Kuhn, Low, Ber. 81, 552 (1948); Kuhn et al., ibid. 83, 448 (1950); Bognar, Makleit, Pharmazie 11, 376 (1956). Yields on partial hydrolysis, besides α-tomatine, the main constituent, β1-, β2-, γ- and δ-tomatine: Kuhn et al., Ber. 90, 203 (1957). α-Tomatine consists of one mol tomatidine linked to a tetrasaccharide composed of 2 mols d-glucose, 1 mol d-xylose and 1 mol d-galactose: Kuhn et al., Angew. Chem. 68, 212 (1956). Proposed as an alternate precipitant to digitonin: Schultz, Sander, Z. Physiol. Chem. 308, 122 (1957). Structure: Reichstein, ibid. 74, 887 (1962). Toxicity study: Wilson et al., Toxicol. Appl. Pharmacol. 3, 39 (1961).