9555. Tosufloxacin
Nomenclature
CAS number: 100490-36-6
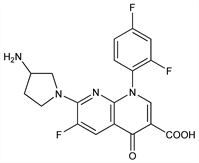
7-(3-Amino-1-pyrrolidinyl)-1-(2,4-difluorophenyl)-6-fluoro-1,4-dihydro-4-oxo-1,8-naphthyridine-3-carboxylic
acid; A-61827. C19H15F3N4O3; mol wt 404.34.
C 56.44%, H 3.74%, F 14.10%, N 13.86%, O 11.87%.
Description and references
Trifluorinated quinolone antibacterial. Prepn: H. Narita et al., DE 3514076 (1985 to Toyama), C.A. 104, 129888r (1986); D. T. W. Chu, EP 153580; idem, US 4616019 (1985, 1986 both to Abbott); and activity: idem et al., J. Med. Chem. 29, 2363 (1986); H. Narita et al., Yakugaku Zasshi 106, 802 (1986), C.A. 106, 196291v (1987). In vitro activity studies of the base: P. B. Fernandes et al., Antimicrob. Agents Chemother. 32, 27 (1988); and in vivo animal studies of the toluenesulfonate: M. Takahata et al., J. Antimicrob. Chemother. 22, 143 (1988). Series of articles on antibacterial activity and clinical evaluation: Chemotherapy (Tokyo) 36, Suppl. 9, 1-1538 (1988).
Derivative
Hydrochloride.Nomenclature
CAS number: 104051-69-6
A-60969. C19H15F3N4O3.HCl; mol wt 440.80.
C 51.77%, H 3.66%, F 12.93%, N 12.71%, O 10.89%, Cl 8.04%.
Properties
Crystals from conc HCl-ethanol (1:3), mp 247-250° (dec).Derivative
Toluenesulfonate.Nomenclature
CAS number: 115964-29-9
Tosufloxacin tosylate; A-64730; T-3262; Ozex (Toyama); Tosuxacin (Dainabot). C19H15F3N4O3.C7H8O3S; mol wt 576.54.
C 54.16%, H 4.02%, F 9.89%, N 9.72%, O 16.65%, S 5.56%.
Properties
Prepd as the monohydrate, mp 258-260°.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Quinolones and Analogs