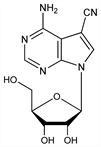
9562. Toyocamycin
Nomenclature
CAS number: 606-58-6
4-Amino-7-β-d-ribofuranosyl-7H-pyrrolo[2,3-d]pyrimidine-5-carbonitrile; 4-amino-5-cyano-7-(d-ribofuranosyl)-7H-pyrrolo[2,3-d]pyrimidine; uramycin B; vengicide; antibiotic 1037; E-212. C12H13N5O4; mol wt 291.26.
C 49.48%, H 4.50%, N 24.05%, O 21.97%.
Description and references
Antibiotic substance extracted from the culture filtrate and mycelium of Streptomyces toyocaensis. Isoln: Nishimura et al., J. Antibiot. 9A, 60 (1956). Structure: Ohkuma, ibid. 14A, 343 (1961). Synthesis of aglycone: Taylor, Hendess, J. Am. Chem. Soc. 86, 951 (1964). Total synthesis: Tolman et al., ibid. 90, 524 (1968); 91, 2102 (1969). Biosynthesis: Uematsu, Suhadolnik, Biochemistry 9, 1260 (1970); eidem, J. Biol. Chem. 245, 4365 (1970). Crystal and molecular structure: P. Prusiner, M. Sundaralingam, Acta Crystallogr. B34, 517 (1978).