9574. Trapidil
Nomenclature
Description and references
First triazolopyrimidine registered as a drug. Prepn: E. Tenor et al., DD 55956 (1967), C.A. 67, 90830f (1967); E. Tenor, R. Ludwig, Pharmazie 26, 534 (1971). Physical and chemical properties: S. Pfeifer et al., ibid. 539. Pharmacology and toxicology: H. Fuller et al., ibid. 554. Metabolic studies: S. Pfeifer et al., ibid. 549; ibid. 27, 752 (1972); I. Bornschein et al., ibid. 33, 51 (1978). Mechanism of action: K. Satoh et al., Arzneim.-Forsch. 30, 1264 (1980). Antiarrhythmic activities in rabbits: M. Sakanashi et al., ibid. 33, 215 (1983). Clinical hemodynamic effects: M. Di Donato et al., ibid. 35, 1295 (1985). GC determn in biological fluids: A. Marzo et al., ibid. 37, 947 (1987).
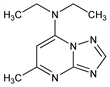
Properties
White to yellowish, odorless and bitter crystalline powder, mp 98-99.4° (Pfeifer); 102-104° from heptane (Tenor). Eutectic temp of mixture with azobenzene: 48°. Very sol in water, 1N sulfuric acid, 10% ammonium hydroxide; easily sol in methanol, isopropanol, n-butanol, chloroform and benzene; sol in ether. Practically insol in hexane, heptane. pKs = 2.79. uv max (methanol): 222, 270, 307 nm (log ε 4.28, 3.83, 4.28). Very stable except under extremely alk conditions. LD50 in mice, rats (mg/kg): 115, 76 i.v.; 380, 235 orally; 155, 100 i.p.; 132, 100 s.c. (Fuller).Derivative
Hydrochloride.Properties
mp 212°.Therapeutic Category
Vasodilator (coronary).
Keywords
Vasodilator (Coronary)