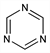
9602. s-Triazine
Nomenclature
CAS number: 290-87-9
1,3,5-Triazine. C3H3N3; mol wt 81.08.
C 44.44%, H 3.73%, N 51.83%.
Description and references
Originally prepd in 1895 and incorrectly identified as "dimeric hydrocyanic acid," C2H2N2. Prepn: C. Grundmann, A. Kreutzberger, J. Am. Chem. Soc. 76, 632, 5646 (1954). Reactivity and use in synthesis of heterocycles: eidem, ibid. 77, 44, 6559 (1955). IR and Raman spectroscopy: J. Goubeau et al., J. Phys. Chem. 58, 1078 (1954). X-ray crystal structure: P. J. Wheatley, Acta Crystallogr. 8, 224 (1955). Determn of enthalpy of combustion and sublimation: K. Bystrm, J. Chem. Thermodyn. 14, 865 (1982). Molecular structure: C. A. Morrison et al., J. Phys. Chem. A 101, 10029 (1997). Review of applications in organic chemistry: T. Güthner, Chim. Oggi 17, 12-14 (1999); G. Giacomelli et al., Curr. Org. Chem. 8, 1497-1519 (2004).