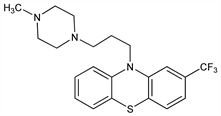
9680. Trifluoperazine
Nomenclature
CAS number: 117-89-5
10-[3-(4-Methyl-1-piperazinyl)propyl]-2-(trifluoromethyl)-10H-phenothiazine; 2-trifluoromethyl-10-[3′-(1-methyl-4-piperazinyl)propyl]phenothiazine. C21H24F3N3S; mol wt 407.50.
C 61.90%, H 5.94%, F 13.99%, N 10.31%, S 7.87%.
Description and references
Prepn: Craig et al., J. Org. Chem. 22, 709 (1957); GB 813861; G. E. Ullyot, US 2921069 (1959 and 1960 to SK & F). Metabolism: T. L. Flanagan et al., J. Pharm. Sci. 51, 996 (1962); C. L. Huang, K. G. Bhansali, ibid. 57, 1511 (1968). Toxicity: P. J. Fowler et al., Arzneim.-Forsch. 27, 866 (1977). Comprehensive description: A. Post et al., Anal. Profiles Drug Subs. 9, 543-581 (1980).
Properties
bp0.6 202-210°. uv max (ethanol): 258, 307.5 nm (log ε 4.50, 3.50). LD50 orally in rats, mice: 542.7, 424.0 mg/ml (Fowler).Derivative
Dihydrochloride.Nomenclature
CAS number: 440-17-5
Triftazin; triphthasine; Eskazinyl (SK & F); Eskazine (GSK); Jatroneural (Procter & Gamble); Modalina (Sanofi-Synthelabo); Stelazine (GSK); Terfluzine (Aventis). C21H24F3N3S.2HCl; mol wt 480.42.
C 52.50%, H 5.45%, F 11.86%, N 8.75%, S 6.67%, Cl 14.76%.
Properties
Cream colored fine powder from abs alc, mp 242-243°. Hygroscopic. Freely sol in water. Insol in dil base, ether, benzene. pK1 3.9, pK2 8.1. pH of 5% aq soln 2.2.Therapeutic Category
Antipsychotic.
Keywords
Antipsychotic; Phenothiazines