9687. Trifluridine
Nomenclature
Description and references
Prepn: C. Heidelberger et al., J. Am. Chem. Soc. 84, 3597 (1962); eidem, J. Med. Chem. 7, 1 (1964); C. Heidelberger, US 3201387 (1965 to U.S. Dept. HEW). Crystal structure: A. H. Tench, Diss. Abstr. Int. B 33, 3587 (1973). NMR study: R. J. Cushley et al., J. Am. Chem. Soc. 90, 709 (1968). Metabolism: D. L. Dexter et al., Cancer Res. 32, 247 (1972); W. J. O'Brien, H. F. Edelhauser, Invest. Ophthalmol. Visual Sci. 16, 1093 (1977). Pharmacodynamics: B. L. Wigdahl, J. R. Parkhurst, Antimicrob. Agents Chemother. 14, 470 (1978); G. J. Smith et al., Biochem. Biophys. Res. Commun. 83, 1538 (1978). Teratogenicity study: M. Itoi et al., Arch. Ophthalmol. 93, 46 (1975). Cytotoxicity and mutagenicity study: E. Huberman, C. Heidelberger, Mutat. Res. 14, 130 (1972). Clinical studies: H. E. Kaufman, Invest. Ophthalmol. Visual Sci. 17, 941 (1978); R. A. Hyndiuk et al., Arch. Ophthalmol. 96, 1839 (1978). Review of mechanism of antiviral activity: C. Heidelberger, Ann. N.Y. Acad. Sci. 255, 317 (1975). Review of pharmacology and therapeutic use: A. A. Carmine et al., Drugs 23, 329-353 (1982).
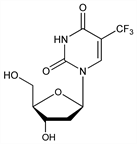
Properties
Cryst from ethyl acetate, mp 186-189°. uv max (0.1N HCl): 260 nm (ε 9960); (0.1N NaOH): 260 nm (ε 6590).Therapeutic Category
Antiviral (ophthalmic).
Keywords
Antiviral; Nucleosides/Nucleotides