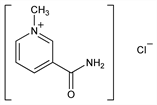
9693. Trigonellamide Chloride
Nomenclature
CAS number: 1005-24-9
3-(Aminocarbonyl)-1-methylpyridinium chloride; N1-methylnicotinamide chloride; 1-methylpyridine-3-carboxylic acid amide chloride; nicotinamide chloromethylate; nicotinamide methyl chloride. C7H9ClN2O; mol wt 172.61.
C 48.71%, H 5.26%, Cl 20.54%, N 16.23%, O 9.27%.
Description and references
One of the principal excretion products of the metabolism of nicotinic acid in man, dog, and rat. Coenzyme action: Warburg, Christian, Biochem. Z. 287, 291 (1936). Synthesis by refluxing nicotinamide with methyl iodide in methanol, then shaking the nicotinamide methiodide with AgCl: Karrer et al., Helv. Chim. Acta 19, 826 (1936); and isolation from urine: Huff, Perizweig, J. Biol. Chem. 150, 395 (1943). Differentiation from NAD: Carpenter, Kodicek, Biochem. J. 46, 421 (1950). In vitro metabolism: G. S. Johnson, Eur. J. Biochem. 112, 635 (1980); H. Hoshino et al., Biochim. Biophys. Acta 801, 250 (1984). HPLC determn in urine: M. A. Kutnink et al., J. Liq. Chromatogr. 7, 969 (1984).