9694. Trigonelline
Nomenclature
CAS number: 535-83-1
3-Carboxy-1-methylpyridinium inner salt; nicotinic acid N-methylbetaine; coffearine; caffearine; gynesine; trigenolline. C7H7NO2; mol wt 137.14.
C 61.31%, H 5.14%, N 10.21%, O 23.33%.
Description and references
In seeds of Trigonella foenumgraecum L., Leguminosae, in coffee beans, in seeds of Strophanthus spp, Apocynaceae and of Cannabis sativa L., Moraceae, in seeds of many other plants; also in sea urchin, Arabacia pustulosa, and in jellyfish, Velella spirans. Excreted in urine after taking nicotinic acid: Ackermann, Z. Biol. 59, 17 (1912). Isoln from normal urine: Linnewah, Renwein, Z. Physiol. Chem. 207, 48 (1932); 209, 110 (1932). Syntheses: Turnau, Monatsh. Chem. 26, 551 (1905); Sarett et al., J. Biol. Chem. 135, 483 (1940); Green, Tong, J. Am. Chem. Soc. 78, 4896 (1956); Kosower, Patton, J. Org. Chem. 26, 1318 (1961). Toxicity study: Brazda, Coulson, Proc. Soc. Exp. Biol. Med. 62, 19 (1946).
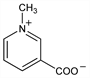
Derivative
Monohydrate.Properties
Crystals from ethanol, mp 230-233°. Salty taste. Very sol in water; sol in alcohol. Practically insol in ether, chloroform. LD50 s.c. in rats: 5.0 g/kg (Brazda, Coulson).Derivative
Hydrochloride.C7H7NO2.HCl; mol wt 173.60.
C 48.43%, H 4.64%, N 8.07%, O 18.43%, Cl 20.42%.