9695. Trihexyphenidyl Hydrochloride
Nomenclature
CAS number: 52-49-3
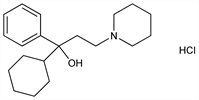
α-Cyclohexyl-α-phenyl-1-piperidinepropanol hydrochloride; 3-(1-piperidyl)-1-cyclohexyl-1-phenyl-1-propanol hydrochloride; 1-phenyl-1-cyclohexyl-3-piperidyl-1-propanol hydrochloride; benzhexol chloride; Aparkane (ICN); Artane (Wyeth); Broflex (Bioglan); Cyclodol; Pacitane (Wyeth); Paralest (Parachemie); Pargitan (Abigo); Parkinane (Wyeth); Parkopan (Hexal); Peragit (AFI); Pipanol (Winthrop); Sedrena (Daiichi); Tremin (Schering-Plough); Triphedinon (Toho); Triphenidyl (Leciva); Tsiklodol. C20H32ClNO; mol wt 337.93.
C 71.08%, H 9.54%, Cl 10.49%, N 4.14%, O 4.73%.
Description and references
Anticholinergic. Synthesis: Denton et al., J. Am. Chem. Soc. 71, 2053 (1949); Adamson, Wilkinson, US 2682543 (1954 to Burroughs Wellcome); Denton, US 2716121 (1955 to Am. Cyanamid). Resolution into isomers: Adamson, Duffin, GB 750156 (1956 to Wellcome Found.).
Properties
Crystals, dec 258.5°. Free base, mp 114.3-115.0°. Soly (g/100 ml): water at 25°, 1.0; alcohol 6; chloroform 5. More sol in methanol; very slightly sol in ether, benzene. pH of a 1% aq soln 5.5-6.0.Derivative
l-Form.Properties
Crystals from isopropyl alc, mp 264°. [α]D20 30° (c = 0.4 in chloroform). Free base, mp 112-113°. [α]D20 25° (c = 0.4 in ethanol).Therapeutic Category
Antiparkinsonian.
Keywords
Antiparkinsonian; Antimuscarinic