9698. Trilostane
Nomenclature
Description and references
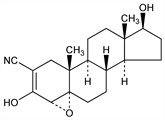
Synthetic steroid analog; competitive inhibitor of 3β-hydroxysteroid dehydrogenase. Prepn: R. O. Clinton, A. J. Manson, US 3296255 (1967 to Sterling); H. C. Neumann et al., J. Med. Chem. 13, 948 (1970). Inhibition of steroid biosynthesis: G. O. Potts et al., Steroids 32, 257 (1978). Disposition in animals: J. F. Baker et al., Arch. Int. Pharmacodyn. Ther. 243, 4 (1980). Metabolism in rats: Y. Mori et al., Chem. Pharm. Bull. 29, 2646 (1981); in humans: D. T. Robinson et al., J. Steroid Biochem. 21, 601 (1984). HPLC determn in plasma: P. Powles et al., J. Chromatogr. 311, 434 (1984); and of major metabolite, 17-ketotrilostane: R. R. Brown et al., ibid. 339, 440 (1985). Experience in treatment of canine Alopecia X: R. Cerundolo et al., Vet. Dermatol. 15, 285 (2004). Clinical study in Cushing's syndrome: P. Komanicky et al., J. Clin. Endocrinol. Metab. 47, 1042 (1978). Review of clinical experience in advanced breast cancer: J. R. Puddefoot et al., Expert Opin. Pharmacother. 7, 2413-2419 (2006).
Properties
Tan crystals from pyridine/dioxane, mp 257.8-270° (dec). [α]D25 +137.4° (c = 1 in pyridine). uv max (ethanol): 252 nm (ε 8300).Therapeutic Category
Adrenocortical suppressant; in treatment of breast cancer.
Therapeutic Category (Veterinary)
Adrenocortical suppressant.Keywords
Adrenocortical Suppressant; Antineoplastic (Hormonal); Antiadrenals