9706. Trimethadione
Nomenclature
Description and references
Synthesis: Spielman, J. Am. Chem. Soc. 66, 1244 (1944); US 2575692 (1951 to Abbott); Davies, Hook, US 2559011 (1951 to Brit. Schering). Metabolism to dimethadione, q.v.: T. C. Butler, J. Pharmacol. Exp. Ther. 108, 11 (1953); T. C. Butler, W. J. Waddell, ibid. 110, 241 (1954). Anticonvulsant activity: H. Ferngren, Acta Pharmacol. Toxicol. 26, 177 (1968); H. H. Frey, ibid. 27, 295 (1969). Pharmacodynamics and metabolism in rats: D. O. Thueson et al., Epilepsia 15, 563 (1974). Evaluation in teratogenesis: J. German et al., Teratology 3, 349 (1970); A. B. Rifkind, Toxicol. Appl. Pharmacol. 30, 452 (1974). GC determn in serum: E. Tanaka, S. Misawa, J. Chromatogr. 413, 376 (1987); LC determn in serum: M. Okamoto et al., Chromatographia 23, 325 (1987). Use in treatment of petit mal epilepsy: S. Livingston et al., J. Am. Med. Assoc. 194, 227 (1965). Use in dissolution of pancreatic stones in humans and dogs: A. Noda et al., Lancet 2, 351 (1984); eidem, Gastroenterology 93, 1002 (1987).
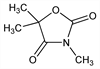
Properties
Granules, crystals, mp 46-46.5°. bp5 78-80°. Slight camphor-like odor. Burning, faintly bitter taste. Soly in water about 5%; increased by the addition of urethan. Freely sol in alcohol, benzene, chloroform, ether. Practically insol in petr ether. The pH of a 5% soln is ≈6.0.Therapeutic Category
Anticonvulsant.
Therapeutic Category (Veterinary)
Anticonvulsant.Keywords
Anticonvulsant