9721. Trimetrexate
Nomenclature
Description and references
Lipophilic dihydrofolate reductase inhibitor structurally related to methotrexate, q.v., with antimicrobial and antitumor activity. Prepn: E. F. Elslager, L. M. Werbel, GB 1345502 (1974 to Parke, Davis); E. F. Elslager et al., J. Med. Chem. 26, 1753 (1983); of water soluble salts: N. L. Colbry, EP 51415; idem, US 4376858 (1982, 1983 both to Warner-Lambert). In vitro antifolate activity and in vivo antitumor effect: J. R. Bertino et al., Biochem. Pharmacol. 28, 1983 (1979). Pharmacology: E. C. Weir et al., Cancer Res. 42, 1696 (1982); R. C. Jackson et al., Adv. Enzyme Regul. 22, 187 (1984). GC-MS determn in human plasma: P. L. Stetson, W. D. Ensminger, J. Chromatogr. 383, 69 (1986). Clinical evaluation with leucovorin vs Pneumocystis carinii in patients with AIDS: C. J. Allegra et al., N. Engl. J. Med. 317, 978 (1987). Review of pharmacology and clinical efficacy: J. T. Lin, J. R. Bertino, J. Clin. Oncol. 5, 2032-2040 (1987); P. J. O'Dwyer et al., NCI Monogr. 5, 105-109 (1987).
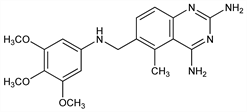
Derivative
Monoacetate monohydrate.Nomenclature
Properties
Crystals from aqueous acetic acid, mp 215-217°. Poorly sol in water. LD50 i.p. in mice: 175 mg/kg (Jackson).Derivative
d-Glucuronate.Nomenclature
Properties
Tan colored solid. Soly in water: >50 mg/ml.Therapeutic Category
Antineoplastic; antipneumocystis.
Keywords
Antineoplastic; Antimetabolites; Folic Acid Analogs/Antagonists