9723. Trimoprostil
Nomenclature
Description and references
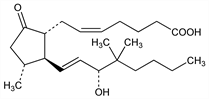
Synthetic prostaglandin E2 analog with antisecretory activity. Prepn: G. W. Holland et al., DE 2437622; eidem, US 4052446; US 4190587 (1975, 1977, 1980 all to Hoffmann-La Roche). Pharmacology: D. E. Wilson, S. L. Winter, Prostaglandins 16, 127 (1978); S. P. Lee et al., Eur. J. Clin. Invest. 17, 1 (1987). Effect on bicarbonate secretion: M. Feldman, J. Clin. Invest. 72, 295 (1983); on inhibition of gastric acid secretion: R. J. Wills et al., Clin. Pharmacol. Ther. 37, 113 (1985). Metabolism: S. J. Kolis et al., Drug Metab. Dispos. 14, 465 (1986). Clinical pharmacokinetics: R. J. Wills et al., J. Clin. Pharmacol. 26, 48 (1986). Clinical evaluation: H. G. Dammann et al., Arzneim.-Forsch. 36, 500 (1986). Multicenter clinical comparison with cimetidine, q.v.: K. D. Bardhan et al., Scand. J. Gastroenterol. 23, 134 (1988); with aldioxa, q.v.: A. Ishimori et al., Acta Ther. 15, 27 (1989). Toxicity study: M. Shimizu et al., Shin'yaku to Rinsho 35, 2199 (1986), C.A. 106, 150180e (1987).
Properties
Colorless oil. [α]D 51.54° (c = 1 in CHCl3). LD50 in mice, rats (mg/kg): 41, 23 orally; 70, 21 i.p.; 68, 29 s.c. (Shimizu).Therapeutic Category
Antiulcerative.
Keywords
Antiulcerative; Prostaglandin/Prostaglandin Analog