9791. Truxillic Acid
Nomenclature
Description and references
Cinnamic acid polymers obtained from the minor alkaloids of cocaine: Liebermann, Ber. 21, 2342 (1888). Five stereoisomers have been obtained: α-, γ-, ε-, peri- and epi-isomers. Stereochemical configurations: Stoermer, Bacher, Ber. 57B, 15-23 (1924).
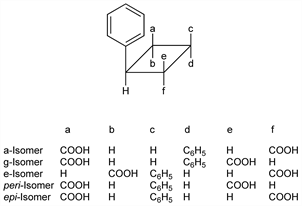
Derivative
α-Isomer.Nomenclature
γ-Isatropaic acid; cocaic acid.Description and references
Prepd by irradiation of cinnamic acid in water: White, Dunathan, J. Am. Chem. Soc. 78, 6055 (1956).
Properties
Crystals from acetic acid, mp 284-285°. Sparingly sol in boiling water, in ether, benzene, carbon disulfide; sol in hot glacial acetic acid, hot alc; sparingly sol in acetone.Derivative
γ-Isomer.Nomenclature
ε-Isatropaic acid.Description and references
γ-Truxillic anhydride, obtained by heating α-truxillic acid with acetic anhydride and sodium acetate, was heated with alkali and the free acid pptd with HCl: Liebermann, Ber. 22, 124 (1889).
Properties
Needles from dil alc, mp 228°. Very slightly sol in hot water; sol in ether.Derivative
ε-Isomer.Nomenclature
β-Cocaic acid.Description and references
Prepd by fusion of α-truxillic acid with KOH: Hesse, Ann. 271, 180 (1892).
Properties
Needles from ether, mp 192°. Freely sol in glacial acetic acid, abs alcohol, chloroform; less sol in benzene. Practically insol in ligroin.Derivative
peri-Isomer.Nomenclature
η-Truxillic acid.Description and references
Prepn: γ-Truxillic anhydride heated at low pressure forms the peri-anhydride, converted to the acid by warming with alcoholic KOH: Stoermer, Bacher, loc. cit.
Properties
Crystals from benzene + ligroin, mp 266° (effervescence). Soluble in alcohol. Practically insol in ether, benzene.Derivative
epi-Isomer.Description and references
Prepd by boiling peri-truxillic acid with excess 10% NaOH: Stoermer, Bacher, loc. cit.