9796. Tryptamine
Nomenclature
CAS number: 61-54-1
1H-Indole-3-ethanamine; 3-(2-aminoethyl)indole; 2-(3-indolyl)ethylamine. C10H12N2; mol wt 160.22.
C 74.96%, H 7.55%, N 17.48%.
Description and references
Occurs in plants. Synthesis starting with nitroethylene and indole: Noland, Hartman, J. Am. Chem. Soc. 76, 3227 (1954). Alternate routes: Thesing, Schulde, Ber. 85, 324 (1952); Jackson, Smith, J. Chem. Soc. 1965, 3498; Tacconi, Farmaco Ed. Sci. 20, 902 (1965); S. Takano et al., Heterocycles 6, 1167 (1977); I. Fleming, M. Woolias, J. Chem. Soc. Perkin Trans. 1 1979, 829. X-ray structure determn: Wakahara et al., Tetrahedron Lett. 1970, 4999. Review of tryptamine syntheses: J. E. Saxton in R. H. F. Manske, The Alkaloids vol. VIII (1965) pp 8-10.
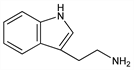
Properties
Needles from petr ether, mp 118°. uv max (ethanol): 222, 282, 290 nm (log ε 4.56, 3.78, 3.71). Sol in ethanol, acetone. Practically insol in water, ether, benzene, chloroform.Derivative
Hydrochloride.C10H12N2.HCl; mol wt 196.68.
C 61.07%, H 6.66%, N 14.24%, Cl 18.03%.