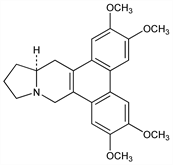
9831. Tylophorine
Nomenclature
Description and references
Major alkaloid from Tylophora asthmatica Wight et Arn., Asclepiadaceae. Also found in other Asclepiadaceae, Moraceae, Urticaceae and Lauraceae. Naturally occurring form originally isolated and reported to be levorotatory; later corrected to dextrorotatory. Isoln: A. N. Ratnagiriswaran, K. Venkatachalam, Indian J. Med. Res. 22, 433 (1935); R. N. Chopra et al., Arch. Pharm. 275, 236 (1937); T. R. Govindachari et al., J. Chem. Soc. 1954, 2801. Structure: eidem, Tetrahedron 9, 53 (1960). Absolute configuration: eidem, J. Chem. Soc. Perkin Trans. 1 1974, 1161. Synthesis of the dl-form: eidem, Chem. Ind. (London) 1960, 664; N. A. Khatri et al., J. Am. Chem. Soc. 103, 6387 (1981). Synthesis and verification of dextrorotation of naturally occurring form: T. F. Buckley 3rd, H. Rapoport, J. Org. Chem. 48, 4222 (1983); J. E. Nordlander, F. G. Njoroge, ibid. 52, 1627 (1987). Stereoselective synthesis of l-form: M. Ihara et al., Tetrahedron Lett. 29, 4135 (1988). Biosynthesis: Mulchandani et al., Phytochemistry 8, 1931 (1969); ibid. 10, 1047 (1971); D. S. Bhakuni, V. K. Mangla, Tetrahedron 37, 401 (1981). Pharmacology: C. Gopalakrishnan et al., Indian J. Med. Res. 69, 513 (1979); 71, 940 (1980).