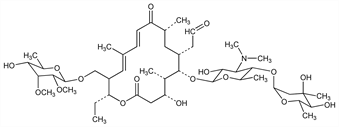
9832. Tylosin
Nomenclature
Description and references
Macrolide antibiotic isolated from a strain of Streptomycetes fradiae found in soil from Thailand: Hamill et al., Antibiot. Chemother. 11, 328 (1961); eidem, US 3178341 (1965 to Lilly). Prodn in batch and chemostat cultures: P. P. Gray, S. Bhuwapathanapun, Biotechnol. Bioeng. 22, 1785 (1980). Partial structure: Morin, Gorman, Tetrahedron Lett. 1964, 2339. Structure: Morin et al., ibid. 1970, 4737; Achenbach et al., Ber. 108, 2481 (1975). Configurational study: S. Omura et al., Tetrahedron Lett. 1977, 1045. Abs config: eidem, J. Antibiot. 33, 915 (1980); N. D. Jones et al., ibid. 35, 420 (1982). Synthesis of tylonolide, the aglycone: S. Masamune et al., J. Am. Chem. Soc. 98, 7874 (1976); K. Tatsuta et al., Tetrahedron Lett. 22, 3997 (1981). Relationship of ribosomal binding and antibacterial properties: J. W. Corcoran et al., J. Antibiot. 30, 1012 (1977). Biosynthesis studies: E. T. Seno et al., Antimicrob. Agents Chemother. 11, 455 (1977); S. Omura et al., J. Antibiot. 31, 254 (1978).