9845. Uliginosins
Description and references
Antibiotics isolated from Hypericum uliginosum HBK, a woody herb found in Mexico and Central America. Isoln: Taylor, Brooker, Lloydia 32, 217 (1969). Structure: Parker, Johnson, J. Am. Chem. Soc. 90, 4716 (1968); Parker et al., ibid. 4723. Synthesis of uliginosin A and dihydrouliginosin B: Meikle, Stevens, Chem. Commun. 1972, 123; eidem, J. Chem. Soc. Perkin Trans. 1 1978, 1303. Synthesis of uliginosin B: eidem, Tetrahedron Lett. 1972, 4787; eidem, J. Chem. Soc. Perkin Trans. 1 1979, 2563.
Derivative
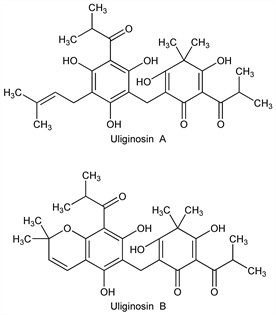
Uliginosin A.Nomenclature
CAS number: 19809-78-0
3,5-Dihydroxy-4,4-dimethyl-2-(2-methyl-1-oxopropyl)-6-[[2,4,6-trihydroxy-3-(3-methyl-2-butenyl)-5-(2-methyl-1-oxopropyl)phenyl]methyl]-2,5-cyclohexadien-1-one. C28H36O8; mol wt 500.58.
C 67.18%, H 7.25%, O 25.57%.
Properties
Pale yellow crystals, mp 160.5-161.5° from acetonitrile-chloroform (4:1). uv max (cyclohexane): 229, 293 nm (ε 31500, 25000).Derivative
Uliginosin B.Nomenclature
CAS number: 19809-79-1
2-[[5,7-Dihydroxy-2,2-dimethyl-8-(2-methyl-1-oxopropyl)-2H-1-benzopyran-6-yl]methyl]-3,5-dihydroxy-4,4-dimethyl-6-(2-methyl-1-oxopropyl)-2,5-cyclohexadien-1-one. C28H34O8; mol wt 498.56.
C 67.45%, H 6.87%, O 25.67%.