10336. Ulipristal Acetate
Nomenclature
Description and references
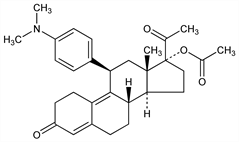
Selective progesterone receptor modulator (SPRM); synthetic 19-norprogesterone derivative. Prepn: C. E. Cook et al., WO 8912448; eidem, US 4954490 (1989, 1990 both to Research Triangle Inst.). Large scale synthesis: P. N. Rao et al., Steroids 65, 395 (2000) DOI PubMed. Pharmacology: E. E. Gainer, A. Ulmann, ibid. 68, 1005 (2003) DOI PubMed; D. L. Blithe et al., ibid., 1013 DOI PubMed. Clinical evaluation as ovulation inhibitor: N. Chabbert-Buffet et al., J. Clin. Endocrinol. Metab. 92, 3582 (2007) DOI PubMed; in treatment of uterine fibroids: E. D. Levens et al., Obstet. Gynecol. 111, 1129 (2008) DOI PubMed. Review: P. A. Orihuela, Curr. Opin. Investig. Drugs 8, 859-866 (2007) PubMed.
Properties
Crystals from aq ethanol, mp 183-185° (Rao); also reported as crystals from methanol + water, mp 118-121° (Cook). uv max (methanol): 261 nm.Derivative
Ulipristal.Nomenclature
Properties
Crystals from ether, mp 125-128°.Therapeutic Category
In treatment of uterine fibroids.
Keywords
Selective Progesterone Receptor Modulator (SPRM)