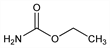
9874. Urethan
Nomenclature
Description and references
Naturally occurring contaminant in fermented foods, particularly wine, stone-fruit brandies, and bread. Prepd by heating urea with alcohol under pressure; by warming urea nitrate with alcohol and sodium nitrite. Toxicity data: K. J. Franklin, J. Pharmacol. Exp. Ther. 42, 1 (1931). Review of carcinogenic action and metabolism: S. S. Mirvish in Adv. Cancer Res. 11, 1-42 (1968); of mutagenicity, metabolism and interactions with DNA: R. E. Sotomayor, T. F. X. Collins, Toxicol. Ind. Health 6, 71-108 (1990); of physiological effects in animals: K. J. Field, C. M. Lang, Lab. Anim. 22, 255-262 (1988). Review of analysis, occurrence and formation in foodstuffs: R. Battaglia et al., Food Addit. Contam. 7, 477-496 (1990); B. Zimmerli, J. Schlatter, Mutat. Res. 259, 325-350 (1991).