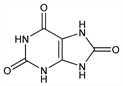
9875. Uric Acid
Nomenclature
CAS number: 69-93-2
7,9-Dihydro-1H-purine-2,6,8(3H)-trione; 8-hydroxyxanthine; purine-2,6,8-triol; purine-2,6,8(1H,3H,9H)-trione; 2,6,8-trioxypurine. C5H4N4O3; mol wt 168.11.
C 35.72%, H 2.40%, N 33.33%, O 28.55%.
Description and references
Discovered by Scheele and independently by Bergman in 1776. It forms the chief end-product of the nitrogenous metabolism of birds and of scaly reptiles and is found in their excrement; present in the urine of all carnivorous animals. Prepn from urea: Bills et al., J. Org. Chem. 27, 4633 (1962). Role in biological processes: Bishop, Talbott, Pharmacol. Rev. 5, 231 (1953).