9889. Ursodiol
Nomenclature
Description and references
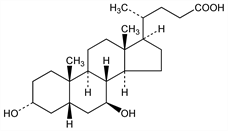
Epimeric with chenodiol, q.v., with respect to the hydroxyl group at C7. Found in bear bile (combined with taurine). Isoln: Shoda, J. Biochem. (Tokyo) 7, 505 (1927). Structure: Kaziro, Z. Physiol. Chem. 185, 151 (1929); 197, 206 (1931); Iwasaki, ibid. 244, 181 (1936). Toxicity data: M. Ardenne, P. G. Reitnauer, Arzneim.-Forsch. 20, 323 (1970). Effect on cholesterol and bile acid metabolism: G. S. Tint et al., Gastroenterology 91, 1007 (1986). Clinical trial in primary biliary cirrhosis: R. E. Poupon et al., N. Engl. J. Med. 324, 1548 (1991); R. Jorgensen et al., Am. J. Gastroenterol. 97, 2647 (2002); in prevention of gallstones following gastric bypass: H. J. Sugerman et al., Am. J. Surgery 169, 91 (1995). Review of pharmacology, toxicology, efficacy: A. Ward et al., Drugs 27, 95-131 (1984). Brief review of clinical effects and comparison with chenodiol: H. Fromm, Gastroenterology 87, 229-233 (1984).
Properties
Bitter plates from alc. mp 203°. [α]D20 +57° (c = 2 in abs ethanol). Freely sol in ethanol, glacial acetic acid; slightly sol in chloroform, sparingly sol in ether. Practically insol in water. LD50 in mice (g/kg): 0.1 i.v. (Ardenne, Reitnauer); in rats, mice (mg/kg): 2000, 6000 s.c.; 1000, 1200 i.p., 310, 260 i.v. (Ward).Derivative
Diformate.Properties
Crystals, mp 170°.Derivative
Diacetate.Properties
Crystals, mp 98-102°.Therapeutic Category
Anticholelithogenic.
Therapeutic Category (Veterinary)
Anticholelithogenic.Keywords
Cholelitholytic Agent