9890. Ursolic Acid
Nomenclature
CAS number: 77-52-1
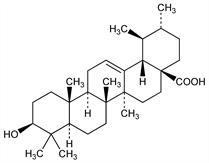
(3β)-3-Hydroxyurs-12-en-28-oic acid; urson; prunol; micromerol; malol. C30H48O3; mol wt 456.70.
C 78.90%, H 10.59%, O 10.51%.
Description and references
In leaves and berries of Arctostaphylos uva-ursi (L.) Spreng (bearberry), of Vaccinium macrocarpon Ait. (cranberry), Rhododendron hymenanthes Makino, Ericaceae. In the protective wax-like coating of apples, pears, prunes, and other fruits. Isoln from apple peelings: Sando, J. Biol. Chem. 56, 457 (1923). Structure: Ruzicka et al., Helv. Chim. Acta 28, 199 (1945); Zurcher et al., ibid. 37, 2145 (1954). Conversion from α-amyrin: Boar et al., J. Chem. Soc. C 1970, 678. Chemistry: Mezzetti et al., Planta Med. 20, 244 (1971).
Properties
Large, lustrous prisms from abs alcohol, fine hair-like needles from dil alcohol, mp 285-288°. [α]D21 +67.5° (c = 1 in N alc KOH). Soly at 15°: One part dissolves in 88 parts methanol, 178 alcohol (35 boiling alcohol), 140 ether, 388 chloroform, 1675 carbon disulfide. Moderately sol in acetone. Sol in hot glacial acetic acid and in 2% alcoholic NaOH. Insol in water and petr ether.Derivative
Acetate.C32H50O4; mol wt 498.74.
C 77.06%, H 10.10%, O 12.83%.
Properties
mp 289-290°. [α]D +62.3° (c = 1.15 in chloroform).Derivative
Methyl ester.C31H50O3; mol wt 470.73.
C 79.10%, H 10.71%, O 10.20%.
Properties
mp 171°. [α]D20 +58° (c = 1.2 in pyridine).Derivative
Methyl ester acetate.C33H52O4; mol wt 512.76.
C 77.30%, H 10.22%, O 12.48%.