9900. Valdecoxib
Nomenclature
Description and references
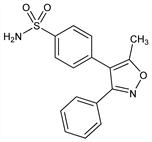
Selective cyclooxygenase-2 (COX-2) inhibitor. Active metabolite of parecoxib, q.v. Prepn: J. J. Talley et al., WO 9625405 (1996 to Searle); eidem, US 5633272 (1997); and activity: eidem, J. Med. Chem. 43, 775 (2000). Chromatographic determn of purity: D. A. Roston et al., J. Pharm. Biomed. Anal. 26, 339 (2001). Gastrointestinal tolerability study: G. M. Eisen et al., Aliment. Pharmacol. Ther. 21, 591 (2005). Clinical trial in hip arthroplasty: F. Camu et al., Am. J. Ther. 9, 43 (2002). Clinical comparison with oxycodone/acetominophen in dental pain: S. E. Daniels et al., J. Am. Dent. Assoc. 133, 611 (2002). Clinical trial in migraine: D. Kudrow et al., Headache 45, 1151 (2005). Review of clinical experience: M. Goldman, S. Schutzer, Formulary 37, 68-77 (2002); of clinical efficacy and safety: G. P. Joshi, Expert Rev. Neurother. 5, 11-24 (2005).
Properties
Crystals, mp 155-157°. Soly at 25°(μg/ml): water 10 (pH 7.0). Sol in methanol, ethanol; freely sol in organic solvents and alkaline (pH = 12) aqueous solns.Therapeutic Category
Anti-inflammatory.
Keywords
Anti-inflammatory (Nonsteroidal); Cyclooxygenase-2 Selective Inhibitor