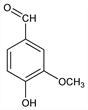
9932. Vanillin
Nomenclature
Description and references
Occurs naturally in a wide variety of foods and plants such as orchids; major commercial source of natural vanillin is from vanilla bean extract. Synthetically produced in-bulk from lignin-based byproducts of paper processes or from guaicol. Known prior to literature identification, first reported in the literature by Bucholtz in 1816 but not isolated until 1858. Isoln from vanilla beans: M. Gobley, Jahresber. Fortschr. Chem. Verw. Theile Anderer Wiss. 1858, 534. LC determn: S. Kahan, D. A. Krueger, J. AOAC Int. 80, 564 (1997). Acute toxicity: P. M. Jenner et al., Food Cosmet. Toxicol. 2, 327 (1964). Brief review of toxicology: D. L. J. Opdyke, ibid. 15, 633-638 (1977). Review of NMR studies: G. J. Martin, Ind. Chem. Libr. 8, 506-527 (1996). Review of synthetic prepn: M. B. Hocking, J. Chem. Educ. 74, 1055-1059 (1997). Reviews: G. S. Clark, Perfum. Flavor. 15, 45-54 (1990); L. J. Esposito et al. in Kirk-Othmer Encyclopedia of Chemical Technology vol. 24 (Wiley-Interscience, New York, 4th ed., 1997) pp 812-825.