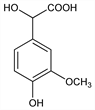
9933. Vanilmandelic Acid
Nomenclature
CAS number: 55-10-7
α,4-Dihydroxy-3-methoxybenzeneacetic acid; 3-methoxy-4-hydroxymandelic acid; 4-hydroxy-3-methoxymandelic acid; VMA. C9H10O5; mol wt 198.17.
C 54.55%, H 5.09%, O 40.37%.
Description and references
Catecholamine metabolite. Urine levels elevated in various pathologies. Misnamed vanillinemandelic acid and vanillylmandelic acid. Prepn from vanillin cyanohydrin: Gardner, Hibbert, J. Am. Chem. Soc. 66, 608 (1944). Improved procedure: Shaw et al., J. Org. Chem. 23, 30 (1958); E. F. Recondo, H. Rinderknecht, ibid. 25, 2248 (1960); I. Goodman et al., Biochem. Prep. 13, 75 (1971). Resolution: Armstrong et al., Biochim. Biophys. Acta 25, 422 (1957). Determination in urine: T. C. Stewart, J. A. Freeman, Vanilmandelic Acid & Catecholamine Determinations (Am. Soc. Clin. Pathol., Chicago, 1976) pp 1-81.