9937. Vasicine
Nomenclature
CAS number: 6159-55-3
1,2,3,9-Tetrahydropyrrolo[2,1-b]quinazolin-3-ol; peganine. C11H12N2O; mol wt 188.23.
C 70.19%, H 6.43%, N 14.88%, O 8.50%.
Description and references
Isoln from Adhatoda vasica Nees, Acanthaceae: Hooper, Pharm. J. 18, 841 (1888); Sen, Ghose, J. Indian Chem. Soc. 1, 315 (1924); Mehta et al., J. Org. Chem. 28, 445 (1963). Isoln from Peganum harmala L., Zygophyllaceae: Spth, Nikawitz, Ber. 67, 45 (1934); Spth, Kuffner, ibid. 67, 868 (1934). Structure and synthesis: Spth et al., ibid. 68, 699 (1935); Spth, Platzer, ibid. 69, 255 (1936). Synthesis of dl-vasicine: Southwick, Casanova, J. Am. Chem. Soc. 80, 1168 (1958). Reviews: Spth, Monatsh. Chem. 72, 115 (1938); H. T. Openshaw, “The Quinazoline Alkaloids” in The Alkaloids vol. III, R. H. F. Manske, H. L. Holmes, Eds. (Academic Press, New York, 1953) pp 101-118; Ray, J. Indian Chem. Soc. 35, 697 (1958).
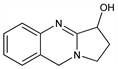
Derivative
dl-Form.Properties
Needles from alc. mp 210°. Sublimes in high vacuum. Sol in acetone, alcohol, chloroform; slightly sol in water, ether, benzene.Derivative
l-Form.Properties
Needles from alc, mp 212°. [α]D14 254° (c = 2.4 in CHCl3); [α]D14 62° (c = 2.4 in alc). In dil HCl this alkaloid is dextrorotatory.Derivative
Hydrochloride dihydrate.Properties
Needles, mp 208° (dry).Derivative
Hydriodide dihydrate.Properties
Needles, mp 195° (dry).Derivative
Methiodide.Properties
Needles from methanol, mp 187°.Derivative
Acetylvasicine.C11H11N2OCOCH3; mol wt 230.26.
C 67.81%, H 6.13%, N 12.17%, O 13.90%.