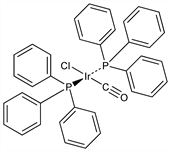
10319. Vaska's Compound
Nomenclature
Description and references
Square-planar iridium complex; homogenous catalyst for a variety of synthetic applications. Model compd for reaction mechanism studies that led to the concepts of oxidative addition and reductive elimination in organotransition metal chemistry. Prepn: L. Vaska, J. W. DiLuzio, J. Am. Chem. Soc. 83, 2784 (1961) DOI. Improved prepn: M. Rahim, K. J. Ahmed, Inorg. Chem. 33, 3003 (1994) DOI. Equilibrium constant determn studies: R. G. Pearson, C. T. Kresge, ibid. 20, 1878 (1981) DOI. Reactivity studies: G. G. Eberhardt, L. Vaska, J. Catal. 8, 183 (1967) DOI; L. Vaska, Acc. Chem. Res. 1, 335 (1968) DOI; C. Douvris, C. A. Reed, Organometallics 27, 807 (2008) DOI. Catalysis applications: Z. Aizenshtat et al., J. Org. Chem. 42, 2386 (1977) DOI; H. A. Zahalka, H. Alper, Organometallics 5, 2497 (1986) DOI; H. Lebel, C. Ladjel, ibid. 27, 2676 (2008) DOI. Brief description and review of synthetic uses: S. A. Westcott, "trans-Carbonyl(chloro)bis(triphenylphosphine)iridium(I)" in Encyclopedia of Reagents for Organic Synthesis 2, L. A. Paquette, Ed. (John Wiley & Sons, New York, 1995) pp 1000-1001.