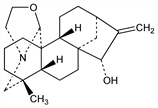
9940. Veatchine
Nomenclature
Description and references
From bark of Garrya veatchii Kellogg, Garryaceae, where it occurs together with garryine and other alkaloids: Oneto, J. Am. Pharm. Assoc. 35, 204 (1946); Wiesner et al., Can. J. Chem. 30, 608 (1952). Structure: Wiesner et al., J. Am. Chem. Soc. 76, 6068 (1954); Pelletier, ibid. 82, 2398 (1960). Stereochemistry: Vorbrüggen, Djerassi, Tetrahedron Lett. 1961, 119; J. Am. Chem. Soc. 84, 2990 (1962). Coexistence of epimers found in crystal and molecular structure: S. W. Pelletier et al., ibid. 100, 7976 (1978). 13C-NMR study of epimers: N. V. Mody, S. W. Pelletier, Tetrahedron 34, 2421 (1978). Racemic syntheses and resolution: Nagata et al., ibid. 86, 929 (1964), 89, 1499 (1967); Guthrie et al., Collect. Czech. Chem. Commun. 31, 602 (1966). Total synthesis of optically active form: Wiesner et al., Experientia 26, 471 (1970).