9968. Verticillins
Description and references
Three antibiotics, verticillins A, B and C, produced by a species of Verticillium (strain TM-759), an imperfect fungus isolated from a basidiocarp of Coltricia cinnamomea (Polystictus cinnamomeus). Isoln, IR, NMR spectra of verticillin A: Katagiri et al., J. Antibiot. 23, 420 (1970); see also Chepenko et al., C.A. 78, 156654n (1973). Structure: Minato et al., Chem. Commun. 1971, 44. Structure of verticillins A, B and C and absolute configuration of verticillins A and B: eidem, J. Chem. Soc. Perkin Trans. 1 1973, 1819. Synthetic studies: Husler, Schmidt, Ber. 107, 2804 (1974); Schmidt et al., ibid. 2816.
Derivative
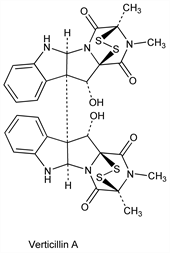
Verticillin A.Nomenclature
CAS number: 32164-16-2
C30H28N6O6S4; mol wt 696.84.
C 51.71%, H 4.05%, N 12.06%, O 13.78%, S 18.41%.
Properties
Pale yellow plates from chloroform, C30H28N6O6S4.CHCl3, mp 199-213° (dec); pale yellow needles from pyridine, C30H28N6O6S4.2/3C5H5N, mp 202-217° (dec); pale yellow amorphous powder from tetrahydrofuran, mp 203-214° (dec). [α]D +703.7° (c = 0.422 in dioxane). uv max (dioxane): 306 nm (ε 5960). LD50 i.p. in mice: 7.6 mg/kg (Katagiri).Derivative
Verticillin B.Nomenclature
CAS number: 52212-86-9
C30H28N6O7S4; mol wt 712.84.
C 50.55%, H 3.96%, N 11.79%, O 15.71%, S 17.99%.
Properties
The mono-3-hydroxymethyl analog of verticillin A. Pale yellow prisms from chloroform, mp 230-233° (dec). [α]D21 +704.7° (c = 0.493 in dioxane). uv max (dioxane): 306 nm (ε 5600).Derivative
Verticillin C.C30H28N6O7S5; mol wt 744.90.
C 48.37%, H 3.79%, N 11.28%, O 15.04%, S 21.52%.