9969. Verticine
Nomenclature
CAS number: 23496-41-5
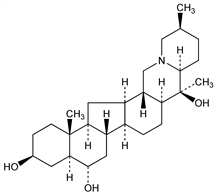
(3β,5α,6α)-Cevane-3,6,20-triol; peimine. C27H45NO3; mol wt 431.65.
C 75.13%, H 10.51%, N 3.24%, O 11.12%.
Description and references
A member of the hexacyclic Ceveratrum group of alkaloids. Isoln from Fritillaria verticillata Willd. var. Thunbergii Baker, Liliaceae: Fukuda: Sci. Rep. Res. Inst. Tohoku Univ. Ser. A 18, 323 (1929); Morimoto, Kimata, Chem. Pharm. Bull. 8, 302 (1960). Also isolated from F. roylei Hook: Chou, Chen, Chin. J. Physiol. 6, 265 (1932), C.A. 26, 5703 (1932). Structure: Ito et al., ibid. 11, 1337 (1963). Abs config: S. Ito et al., Tetrahedron Lett. 1968, 5373. Total synthesis: J. P. Kutney et al., J. Am. Chem. Soc. 99, 964 (1977).
Properties
Needles from ethanol, mp 223-224°. [α]D16 19.4° (ethanol); [α]D17 20° (CHCl3). pK′a 9.5. uv max (ethanol + HCl): 215 nm (ε 10).Derivative
Hydrochloride.C27H45NO3.HCl; mol wt 468.11.
C 69.28%, H 9.90%, N 2.99%, O 10.25%, Cl 7.57%.
Properties
Prisms from water, dec 291-294°. [α]D18 18.5°.Derivative
Perchlorate.C27H45NO3.HClO4; mol wt 532.11.
C 60.94%, H 8.71%, N 2.63%, O 21.05%, Cl 6.66%.
Properties
Prisms from water, dec 273°. [α]D23 15° (c = 0.05).Derivative
Methiodide.C27H45NO3.CH3I; mol wt 573.59.
C 58.63%, H 8.43%, N 2.44%, O 8.37%, I 22.12%.
Properties
Crystals from butanol + ether, dec 205-210°. [α]D18 14.0° (water). Sol in water, methanol, ethanol; practically insol in chloroform, benzene, acetone.Derivative
3-Glucoside.Nomenclature
Peiminoside.C33H55NO8; mol wt 593.79.
C 66.75%, H 9.34%, N 2.36%, O 21.56%.