9981. Viminol
Nomenclature
CAS number: 21363-18-8
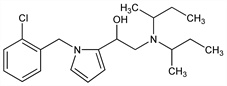
α-[[Bis(1-methylpropyl)amino]methyl]-1-[(2-chlorophenyl)methyl]-1H-pyrrole-2-methanol; 1-(o-chlorobenzyl)-α-[(di-sec-butylamino)methyl]pyrrole-2-methanol; 1-[α-(N-o-chlorobenzyl)pyrryl]-2-di-sec-butylaminoethanol; diviminol; Z-424. C21H31ClN2O; mol wt 362.94.
C 69.49%, H 8.61%, Cl 9.77%, N 7.72%, O 4.41%.
Description and references
Prepn: U. M. Teotino, D. Della Bella, ZA 6702732, eidem, US 3539589 (1970 to Whitefin Holding). Synthesis and metabolic fate of 14C-viminol: Cameron et al., Arzneim.-Forsch. 23, 708 (1973). Viminol possesses 3 asymmetric carbon atoms and thus is composed of 6 stereoisomers. Role of stereoisomers in determining specific pharmacological effects: D. Della Bella et al., Nature New Biol. 241, 282 (1973). Additional pharmacology data: M. Babbini et al., Eur. J. Pharmacol. 23, 137 (1973). Review: D. Della Bella, Boll. Chim. Farm. 111, 5-19 (1972).
Properties
bp0.1 mm 160-165°.Derivative
p-Hydroxybenzoate.Nomenclature
CAS number: 23784-10-3
Dividol (Zambon). C21H31ClN2O.C7H6O3; mol wt 501.06.
C 67.12%, H 7.44%, Cl 7.08%, N 5.59%, O 12.77%.
Properties
mp 128-130° (dec). LD50 i.p. in rats: 205.9 mg/kg (Babbini).Therapeutic Category
Analgesic.