9987. Vindesine
Nomenclature
Description and references
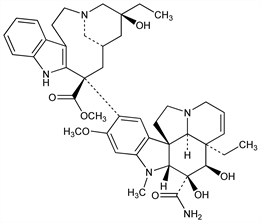
Synthetic deriv of the dimeric Catharanthus alkaloid vinblastine, q.v. Prepn: G. J. Cullinan, K. Gerzon, DE 2415980 (1974 to Lilly), C.A. 82, 72191r (1975); see also C. J. Burnett et al., J. Med. Chem. 21, 88 (1978). 13C-NMR spectroscopy: D. E. Dorman, J. W. Paschal, Org. Magn. Reson. 8, 413 (1976). Antitumor activity in rodents: M. J. Sweeney et al., Cancer Res. 38, 2886 (1978). Pharmacokinetics: R. J. Owellen et al., ibid. 37, 2603 (1977). Use in acute lymphocytic leukemia of childhood: W. Krivit et al., Cancer Chemother. Pharmacol. 2, 267 (1979). Brief review of preclinical and early clinical data: R. W. Dyke et al., ibid. 229. Toxicology: G. C. Todd et al., Toxicol. Environ. Health 1, 843 (1976); R. J. Owellen et al., Biochem. Pharmacol. 26, 1213 (1977).
Properties
Crystals from ethanol-methanol, mp 230-232°. [α]D25 +39.4° (c = 1.0 in methanol). uv max (methanol): 214, 266, 288, 296 nm (ε 53400, 17450, 13950, 12500). pKa′ (DMF 66%) 5.39, 7.36; (H2O) 6.04, 7.67.Derivative
Sulfate salt.Nomenclature
Properties
Amorphous solid from ethanol-isopropyl alcohol, mp >250° (solvated form). LD50 in mice, rats (mg/kg): 6.3 ± 0.6, 2.0 ± 0.2 i.v. (Todd); in mice: 8.8 ± 2.5 i.p. (Owellen, Biochem. Pharmacol.).Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Alkaloids/Natural Products; Vinca Alkaloids