9988. Vindoline
Nomenclature
Description and references
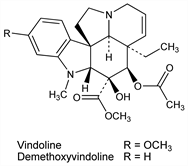
Major alkaloid from the leaves of Vinca rosea Linn. (Catharanthus roseus G. Don.), Apocynaceae; occurs naturally as the ()-form: Gorman et al., J. Am. Pharm. Assoc. 48, 256 (1959); Svoboda et al., ibid. 659; Moza, Trojánek, Collect. Czech. Chem. Commun. 28, 1419 (1963). Structure: Gorman et al., J. Am. Chem. Soc. 84, 1058 (1962); Neuss, Bull. Soc. Chim. Fr. 1963, 1509. Stereochemistry: Moncrief, Lipscomb, J. Am. Chem. Soc. 87, 4963 (1965). Review of chemistry: Neuss et al., Adv. Chemother. 1, 133 (1964). Total synthesis of (±)-vindoline: Ando et al., J. Am. Chem. Soc. 97, 6880 (1975); Y. Ban et al., Tetrahedron Lett. 1978, 151; J. P. Kutney et al., J. Am. Chem. Soc. 100, 4220 (1978). Lacks physiological activity alone but is contained as the pentacyclic moiety in the antineoplastic agents vinblastine and vincristine, q.q.v.
Properties
Needles from acetone + petr ether, mp 164-165°; prisms, mp 174-175°; [α]D20 18° (chloroform); pKa 5.5 in 66% DMF (Moza, Trojánek). Also reported as crystals, mp 154-155°; [α]D27 +42° (chloroform) (Gorman). uv max (ethanol): 212, 250, 304 nm (log ε 4.49, 3.74, 3.57).Derivative
Hydrochloride.Properties
Crystals from acetone, mp 161-164°.Derivative
Demethoxyvindoline.Nomenclature
Description and references
Structure: Moza, Trojánek, Collect. Czech. Chem. Commun. 28, 1427 (1963).