10005. Viridicatin
Nomenclature
CAS number: 129-24-8
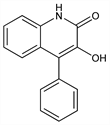
3-Hydroxy-4-phenyl-2(1H)-quinolinone; 3-hydroxy-4-phenylcarbostyril; 2,3-dihydroxy-4-phenylquinoline. C15H11NO2; mol wt 237.25.
C 75.94%, H 4.67%, N 5.90%, O 13.49%.
Description and references
Antibiotic substance from the mycelium of Penicillium viridicatum Westling, the chief mold on stored corn: Cunningham, Freeman, Biochem. J. 53, 328 (1953); from various strains of P. cyclopium Westling: Bracken et al., ibid. 57, 587 (1954); from P. puberulum Bainier: Austin, Meyers, J. Chem. Soc. 1964, 1197. Biosynthesis: Luckner, Tetrahedron Lett. 1962, 1035; Luckner, Mothes, Arch. Pharm. 296, 18 (1963); Framm et al., Eur. J. Biochem. 37, 78 (1973). Synthesis: Eistert, Selzer, Z. Naturforsch. 17b, 202 (1962). Mass spectra: Luckner et al., Tetrahedron 25, 2575 (1969). Antibacterial activity and inhibitory effect on plant growth: Taniguchi, Satomura, Agric. Biol. Chem. 34, 506 (1970).
Properties
Lustrous needles from methanol or ethanol, mp 268°. Sublimes unchanged in high vacuum at 160-170°. Very weak acid. uv and ir curves: Cunningham, Freeman, loc. cit. Practically insol in water, aq NaHCO3. Sparingly sol in cold organic solvents, cold concd HCl, dil mineral acids; sol in cold, aq 2N KOH, glacial acetic acid.Derivative
Sodium salt.C15H10NNaO2; mol wt 259.24.
C 69.50%, H 3.89%, N 5.40%, Na 8.87%, O 12.34%.
Properties
Prismatic needles from aq NaOH, dec 260-265°. Sol in water.Derivative
Monoacetylviridicatin.C17H13NO3; mol wt 279.29.
C 73.11%, H 4.69%, N 5.02%, O 17.19%.
Properties
Crystals from aq ethanol, mp 200-201°. Dec slowly on standing.Derivative
O,O-Dimethylviridicatin.C17H15NO2; mol wt 265.31.
C 76.96%, H 5.70%, N 5.28%, O 12.06%.
Properties
Plates from ethanol, mp 86-87°.Derivative
O,N-Dimethylviridicatin.C17H15NO2; mol wt 265.31.
C 76.96%, H 5.70%, N 5.28%, O 12.06%.