10006. Viridin
Nomenclature
CAS number: 3306-52-3
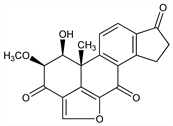
1β-Hydroxy-2β-methoxy-18-norandrosta-5,8,11,13-tetraeno[6,5,4-bc]furan-3,7,17-trione. C20H16O6; mol wt 352.34.
C 68.18%, H 4.58%, O 27.25%.
Description and references
Antibiotic substance from Gliocladium virens: Grove et al., J. Chem. Soc. 1965, 3803. Previously reported isolation from Trichoderma viride: Brian, McGowan, Nature 156, 144 (1945); Brian et al., Ann. Appl. Biol. 33, 190 (1946). Epimerizes to β-viridin where methoxyl group is equatorial. Separation of the two isomers: Vischer et al., Nature 165, 528 (1950). Structure: Grove et al., Chem. Commun. 1965, 343. Configuration: eidem, J. Chem. Soc. C 1966, 743; Neidle, Hursthouse, J. Chem. Soc. Perkin Trans. 2 1972, 760. Biosynthesis: Blight et al., Chem. Commun. 1968, 1117; Grove, J. Chem. Soc. C 1969, 549.
Properties
Prisms from benzene, mp 245° (dec). [α]D19 224°. uv max: 242, 300 nm (log ε 4.49, 4.22). Needles from acetone, mp 222-224° (dec). Prisms from glacial acetic acid, mp 200-205° (dec). Sol in water, chloroform; sparingly sol in carbon disulfide, carbon tetrachloride. Practically insol in ether, camphor. Aq solns lose activity rapidly unless acidified to pH 3.Derivative
β-Isomer.Properties
Prisms from benzene, mp 240-245° (dec). [α]D16 23°. uv max: 243, 300 nm (log ε 4.45, 4.25).Therapeutic Category
Antifungal.
Keywords
Antifungal (Antibiotics)