10018. Vitamin D2
Nomenclature
Description and references
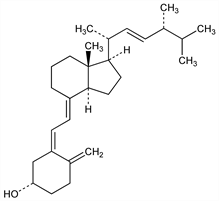
The synthetic form of vitamin D. Prepd from ergosterol by uv irradiation in a suitable solvent: Askew et al., Proc. R. Soc. London B109, 488 (1931/32). The best wavelengths for production of vitamin D2 seem to be from 275 to 300 nm: Knudson, Benford, J. Biol. Chem. 124, 287 (1938); Bunker et al., J. Am. Chem. Soc. 62, 508 (1940). Prepn by electron bombardment of ergosterol using longer waves: AT 119210. Manuf: US 1902785 (1933); US 2030792 (1936); US 1680818 (1928); US 1871136 (1932). Direct total synthesis: B. Lythgoe et al., Tetrahedron Lett. 1977, 3685. Discussion of structural problems: Papers by Askew and Windaus et al., beginning in 1930. Stereochemistry: Crowfoot, Dunitz, Nature 162, 608 (1948); Chem. Ind. (London) 1957, 1149. General review: Inhoffen, Angew. Chem. 72, 875 (1960). A polar, biologically active metabolite of vitamin D2, 25-hydroxyergocalciferol, which is ≈1.5 times more active in curing rickets in rats, has been isolated from pig plasma. See Suda et al., Biochem. Biophys. Res. Commun. 35, 182 (1969); eidem, Biochemistry 8, 3515 (1969).
Properties
Prisms from acetone, mp 115-118°. Sublimes in very high vacuum (0.0006 mm) without dec. [α]D25 +82.6° (c = 3 in acetone); [α]D20 +102.5° (alcohol); [α]D20 +52° (CHCl3). uv max (hexane): 264.5 nm (E1%1cm 458.9 ± 7.5). Not precipitated by digitonin (diff from ergosterol). Insol in water. Sol in the usual organic solvents. 1 ml acetone dissolves 0.0695 g at 7°. Slightly sol in vegetable oils. Commercial solns are usually made with propylene glycol or sesame oil. Vitamin D2 crystals have a potency of 40 units of vitamin D (U.S.P.) per μg. The behavior in storage is described under vitamin D3. See also Huber, Barlow, J. Biol. Chem. 149, 125 (1943) for stability information.Use
Rodenticide.Therapeutic Category
Vitamin (antirachitic).
Therapeutic Category (Veterinary)
Nutritional factor (antirachitic). Low activity in poultry.Keywords
Vitamin/Vitamin Source; Vitamin D