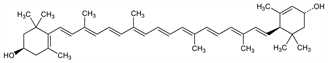
10063. Xanthophyll
Nomenclature
Description and references
One of the most widespread carotenoid alcohols in nature. Originally isolated from egg yolk, also isolated by chromatography from nettles, algae, and petals of many yellow flowers. Occurs also in colored feathers of birds: Volker, Z. Physiol. Chem. 288, 20 (1951). Extraction from petals of Tagetes patula L., Compositae: Karrer et al., Helv. Chim. Acta 30, 531 (1947). Occurs together with zeaxanthin, q.v. Dipalmitate occurs in Helenium autumnale L., Compositae and other flowers: Kuhn, Winterstein, Naturwissenschaften 18, 754 (1930). Conversion to zeaxanthin with sodium alcoholate: Karrer, Jucker, ibid. 266. Does not possess vitamin A potency: Schumacher et al., Poult. Sci. 23, 529 (1944). Stereochemistry: Zechmeister, Chem. Rev. 34, 267 (1944). Structure: Karrer, Helv. Chim. Acta 34, 2160 (1951). Abs config: Goodfellow et al., Chem. Commun. 1970, 1578; Buchecker et al., Chimia 25, 192 (1971); eidem, Helv. Chim. Acta 57, 631 (1974). Synthesis: H. Mayer, A. Rüttimann, ibid. 63, 1451 (1980). Sepn and determn of configurational isomers: A. Rüttiman et al., J. High Resolut. Chromatogr. Chromatogr. Commun. 6, 612 (1983). Reviews: Zechmeister, Carotinoide (Berlin, 1934); Mayer, The Chemistry of Natural Coloring Matters (New York, 1943); Karrer, Jucker, Carotenoids (New York, 1950).