10071. Xenbucin
Nomenclature
CAS number: 959-10-4
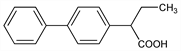
α-Ethyl-[1,1′-biphenyl]-4-acetic acid; 2-(4-biphenylyl)butyric acid; α-(4-biphenylyl)butyric acid; α-(4-diphenylyl)butyric acid; α-(p-xenyl)butyric acid; 4-diphenylylethylacetic acid; 4-biphenylylethylacetic acid; ethyl-p-xenylacetic acid; α-ethyl-p-phenyl-α-toluic acid; MG-1559; Liosol (Maggioni). C16H16O2; mol wt 240.30.
C 79.97%, H 6.71%, O 13.32%.
Description and references
Prepn: Blicke, Grier, J. Am. Chem. Soc. 65, 1725 (1943); Cavallini, Massarani, Farmaco Ed. Sci. 11, 167 (1956). Fibrinolytic activity: R. J. Gryglewski, M. Eckstein, Nature 214, 626 (1967).
Properties
Crystals from glacial acetic acid, mp 123-125°. Soluble in ethanol, methanol, acetone, carbon tetrachloride, benzene, ether, dimethylformamide. Practically insol in water.Derivative
trans-4-Phenylcyclohexylamine salt (1:1).Nomenclature
CAS number: 19992-80-4
Butixirate; MG-5771; Flectar (Maggioni). C28H33NO2; mol wt 415.57.
C 80.92%, H 8.00%, N 3.37%, O 7.70%.
Description and references
Prepn, analgesic and anti-inflammatory properties: G. Cantarelli et al., Farmaco Ed. Sci. 24, 140 (1969); GB 1168542 (1969 to Maggioni), C.A. 72, 43197 (1970). Therapeutic effectiveness: P. Croce, Clin. Ter. 50, 241 (1969).
Properties
Crystals, mp 224-227°. LD50 i.p. in mice: 183 μM/kg (Cantarelli).Therapeutic Category
Free acid as antilipemic; trans-4-phenylcyclohexylamine salt as analgesic, anti-inflammatory.