10190. Zolpidem
Nomenclature
Description and references
Selective non-benzodiazepine GABAA receptor agonist. Prepn: J. P. Kaplan, P. George, EP 50563; eidem, US 4382938 (1982, 1983 both to Synthelabo). Neuropharmacology: H. Depoortere et al., J. Pharmacol. Exp. Ther. 237, 649 (1986). Neurochemical profile: B. Scatton et al., ibid. 659. Binding study in rat brain: S. Arbilla et al., Eur. J. Pharmacol. 130, 257 (1986). HPLC determn in plasma: P. Guinebault et al., J. Chromatogr. 383, 206 (1986); P. R. Ring, J. M. Bostick, J. Pharm. Biomed. Anal. 22, 495 (2000). Review of pharmacology and pharmacokinetics: H. D. Langtry, P. Benfield, Drugs 40, 291-313 (1990); of abuse and dependence potential: G. Hajak et al., Addiction 98, 1371-1378 (2003). Review of clinical experience in insomnia: T. S. Harrison, G. M. Keating, CNS Drugs 19, 65-89 (2005); of extended release formulation: R. L. Barkin, Am. J. Ther. 14, 299-305 (2007).
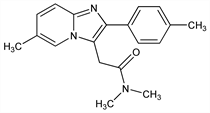
Properties
mp 196°. pKa 6.2. Log P (octanol/water): 2.43.Derivative
l-(+)-Hemitartrate.Nomenclature
Properties
White to off-white crystalline powder. Soly in water (20°): 23 mg/ml. Sparingly sol in alcohol, propylene glycol.Note
This is a controlled substance (depressant): 21 CFR, 1308.14.Therapeutic Category
Sedative, hypnotic.
Keywords
Sedative/Hypnotic; Imidazopyridines