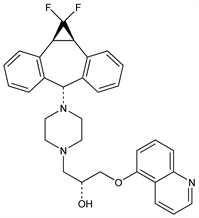
10196. Zosuquidar
Nomenclature
Description and references
Multidrug resistance (MDR) modulator; selective inhibitor of P-glycoprotein (P-gp), q.v. Prepn: J. R. Pfister, D. L. Slate, WO 9424107; eidem, US 5654304 (1994, 1997 both to Syntex); J. R. Pfister et al., Bioorg. Med. Chem. Lett. 5, 2473 (1995). Improved synthesis: C. J. Barnett et al., J. Org. Chem. 69, 7653 (2004). Effect on multidrug resistant cell lines: L. J. Green et al., Biochem. Pharmacol. 61, 1393 (2001). Specificity for P-gp: R. L. Shepard et al., Int. J. Cancer 103, 121 (2003). Clinical pharmacology and pharmacokinetics in patients with advanced malignancies: E. H. Rubin et al., Clin. Cancer Res. 8, 3710 (2002). Effect on pharmacokinetics of doxorubicin: S. Callies et al., Cancer Chemother. Pharmacol. 51, 107 (2003).
Derivative
Trihydrochloride.Nomenclature
Properties
mp 190°.Therapeutic Category
Antineoplastic adjunct (chemosensitizer).
Keywords
Antineoplastic Adjunct; Chemosensitizer