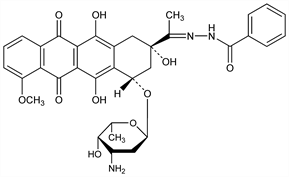
10195. Zorubicin
Nomenclature
CAS number: 54083-22-6
(2S-cis)-Benzoic acid [1-[4-[(3-amino-2,3,6-trideoxy-α-l-lyxo-hexopyranosyl)oxy]-1,2,3,4,6,11-hexahydro-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-2-napthacenyl]ethylidene]hydrazide; benzoic acid hydrazide 3-hydrazone with daunorubicin; RP-22050. C34H35N3O10; mol wt 645.66.
C 63.25%, H 5.46%, N 6.51%, O 24.78%.
Description and references
Semi-synthetic antibiotic related to daunorubicin, q.v. Prepn: G. Jolles, DE 2327211 (1974 to Rhone-Poulenc), C.A. 82, 171381x (1975). Biological activity: R. Maral et al., C. R. Seances Acad. Sci. Ser. D 275, 301 (1972); R. Maral, Cancer Chemother. Pharmacol. 2, 31 (1979). Distribution and metabolism in mice: R. Baurain et al., ibid. 37. Mechanism of action: G. P. Sartiano et al., J. Antibiot. 32, 1038 (1979). Acute cardiovascular effects in dogs: E. H. Herman, R. S. Young, Cancer Treat. Rep. 63, 1771 (1979). Clinical study in breast cancer: J. N. Ingle, ibid. 1701.
Derivative
Hydrochloride.Nomenclature
CAS number: 36508-71-1
NSC-164011; Rubidazone (Bellon). C34H35N3O10.HCl; mol wt 682.12.
C 59.87%, H 5.32%, N 6.16%, O 23.46%, Cl 5.20%.
Properties
Red-orange crystalline powder from ethanol. [α]D20 50° (c = 0.2 in water). uv max (methanol): 232.5, 253, 480, 495 nm (ε 40225, 35300, 10480, 10300). LD50 in mice (mg/kg): 13.66 s.c., 4.42 i.p., 8.50 i.v. (Maral, 1972).Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antibiotics and Analogs; Anthracyclines