10227. Doripenem
Nomenclature
Description and references
Broad spectrum 1β-methyl-carbapenem antibiotic. Prepn: Y. Nishitani et al., EP 528678; eidem, US 5317016 (1993, 1994 both to Shionogi). Synthesis and structure-activity relationship: Y. Iso et al., J. Antibiot. 49, 199 (1996) PubMed. Large-scale synthesis: Y. Nishino et al., Org. Process Res. Dev. 7, 846 (2003) DOI. HPLC determn in serum: C. Sutherland, D. P. Nicolau, J. Chromatogr. B 853, 123 (2007) DOI PubMed. Comparative stability against human recombinant dehydropeptidase-I (DHP-I) versus imipenem and biapenem: M. Mori et al., J. Antimicrob. Chemother. 37, 1034 (1996) DOI. In vitro and in vivo activity: M. Tsuji et al., Antimicrob. Agents Chemother. 42, 94 (1998) PubMed. Comparative antimicrobial spectrum in vitro: T. R. Fritsche et al., Clin. Microbiol. Infect. 11, 974 (2005) DOI PubMed. Activity against carbapenem-resistant isolates of Pseudomonas aeruginosa: S. Sakyo et al., J. Antibiot. 59, 220 (2006) PubMed. Pharmacokinetics: T. Hori et al., In Vivo 20, 91 (2006) PubMed.
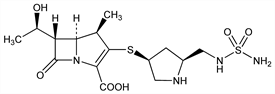
Properties
Colorless foam.Derivative
Hydrate.Nomenclature
Properties
Crystalline powder. Sol in water.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Carbapenems