10244. Bevirimat
Nomenclature
Description and references
HIV maturation inhibitor derived from the natural product, betulinic acid, q.v. Disrupts processing of HIV Gag protein and blocks the conversion to mature capsid. Prepn: K.-H. Lee et al., WO 9639033 (1996 to UNC Chapel Hill); eidem, US 5679828 (1997 to Biotech; UNC Chapel Hill); Y. Kashiwada et al., J. Med. Chem. 39, 1016 (1996) DOI PubMed. Anti-HIV activity: T. Kanamoto et al., Antimicrob. Agents Chemother. 45, 1225 (2001) DOI PubMed. Mechanism of action: F. Li et al., Proc. Natl. Acad. Sci. USA 100, 13555 (2003) DOI PubMed; eidem, Virology 356, 217 (2006) DOI PubMed. Clinical pharmacokinetics: D. E. Martin et al., Antimicrob. Agents Chemother. 51, 3063 (2007) DOI PubMed. Review: Z. Temesgen, J. E. Feinberg, Curr. Opin. Investig. Drugs 7, 759-765 (2006) PubMed.
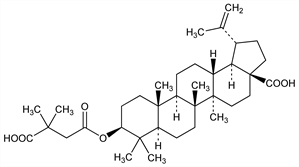
Properties
Colorless needles from methanol, mp 274-276°. [α]D19 +23.5° (c = 0.71 in chloroform/methanol).Derivative
Dimeglumine salt.Nomenclature
Description and references
Prepn: M. D. Power, D. E. Martin, WO 05090380; eidem, US 050239748 (both 2005 to Panacos).
Properties
White solid from diethyl ether. Soly (mg/ml): propylene glycol >255, methanol 37.03, ethanol 24.99, deionized water >7.Therapeutic Category
Antiretroviral.
Keywords
Antiretroviral; Maturation Inhibitors