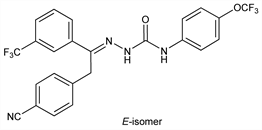
10290. Metaflumizone
Nomenclature
Description and references
Semicarbazone insecticide dervied from pyrazoline that blocks voltage-dependent sodium channels. Prepn: K. Takagi et al., EP 462456; eidem, US 5543573 (1991, 1996 both to Nihon Nohyaku). Laboratory trial against Colorado potato beetle: G. C. Cutler et al., Resistant Pest Manag. Newsl. 16, 33 (2006). Field trial for control of red imported fire ants: X. P. Hu, D. Song, Sociobiology 50, 1107 (2007); against beet armyworm in tomato: J. E. Taylor, D. G. Riley, J. Entomol Sci. 42, 430 (2007). Series of articles on discovery, development, mode of action, efficacy, and pharmacokinetics for veterinary use: Vet. Parasitol. 150, 175-281 (2007) DOI. Toxicological profile: K. Hempel et al., ibid., 190 DOI PubMed. Efficacy of spot-on formulation against KS1 flea strain in cats: M. Dryden et al., ibid. 151, 74 (2008) DOI PubMed.