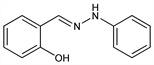
10306. Salicylaldehyde Phenylhydrazone
Nomenclature
Description and references
Indicator used for the titration of Grignard, organolithium, and hydridic reagents. Prepn from salicylaldehyde and phenylhydrazine: E. Fischer, Ber. 17, 572 (1884); A. Rssing, ibid. 2988. Formation mechanism and kinetics: M. P. Bastos et al., J. Org. Chem. 46, 3342 (1981) DOI. Spectroscopy studies: M. H. M. Abou-El-Wafa, U. M. Rabie, Rev. Roum. Chim. 46, 723 (2001); H. H. Hammud et al., Spectrochim. Acta A 63, 255 (2006) DOI PubMed. Polarography studies: V. Krishnaswamy, M. Anbu, Acta Cienc. Indica Chem. 17C, 387 (1991). Tautomerism: G. N. Ledesma et al., J. Mol. Struct. 415, 115 (1997) DOI; and metal coordination properties: idem et al., Polyhedron 17, 1517 (1998) DOI; G. A. Ibaez et al., J. Mol. Struct. 605, 17 (2002) DOI. Metal chelate studies: P. Umpathy et al., Indian J. Chem. 3, 471 (1965); N. Thankarajan, K. P. Kumar, ibid. 30A, 646 (1991); eidem, J. Indian Chem. Soc. 69, 780 (1992). Organometallic reagent titrations: B. E. Love, E. G. Jones, J. Org. Chem. 64, 3755 (1999) DOI PubMed.