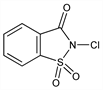
10329. N-Chlorosaccharin
Nomenclature
CAS number: 14070-51-0
2-Chloro-1,2-benzisothiazol-3(2H)-one 1,1-dioxide; o-benzoic N-chlorosulphinide; NCSA; NCSac. C7H4ClNO3S; mol wt 217.63.
C 38.63%, H 1.85%, Cl 16.29%, N 6.44%, O 22.05%, S 14.73%.
Description and references
Reagent that is a source of electrophilic chlorine; utilized for a variety of chlorination and oxidation reactions. Prepn: F. D. Chattaway, J. Chem. Soc. Trans. 87, 1882 (1905) DOI. Green prepn: S. P. L. de Souza et al., Synth. Commun. 33, 935 (2003) DOI. Stability and chlorine potential studies: H.-S. Dawn et al., J. Pharm. Sci. 59, 955 (1970) DOI PubMed. Synthetic applications: J. M. Bachhawat et al., Indian J. Chem. 11, 609 (1973); D. Dolenc, B. Sket, Synlett 1995, 327 DOI; N. Iranpoor et al., Can. J. Chem. 84, 69 (2006) DOI. Oxidimetric titration studies: N. Jayasree, P. Indrasenan, Indian J. Chem. 26A, 714 (1987). Review: P. E. Gama, Synlett 2008, 1742-1743 DOI.