2163. Chloroquine
Nomenclature
Description and references
Prepd by the condensation of 4,7-dichloroquinoline with 1-diethylamino-4-aminopentane: DE 683692 (1939); H. Andersag et al., US 2233970 (1941 to Winthrop); Surrey, Hammer, J. Am. Chem. Soc. 68, 113 (1946). Review: Hahn in Antibiotics vol. 3, J. W. Corcoran, F. E. Hahn, Eds. (Springer-Verlag, New York, 1975) pp 58-78. Comprehensive description: D. D. Hong, Anal. Profiles Drug Subs. 5, 61-85 (1976). Comparative clinical trial with dapsone in rheumatoid arthritis: P. D. Fowler et al., Ann. Rheum. Dis. 43, 200 (1984); with penicillamine: T. Gibson et al., Br. J. Rheumatol. 26, 279 (1987).
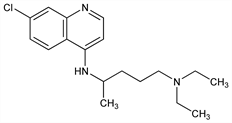
Properties
mp 87°.Derivative
Diphosphate.Nomenclature
Properties
Bitter, colorless crystals. Dimorphic. One modification, mp 193-195°; the other, mp 215-218°. Freely sol in water; pH of 1% soln about 4.5; less sol at neutral and alkaline pH. Stable to heat in solns of pH 4.0 to 6.5. Practically insol in alcohol, benzene, chloroform, ether.Derivative
Sulfate.Nomenclature
Therapeutic Category
Antimalarial; antiamebic; antirheumatic. Lupus erythematosus suppressant.
Keywords
Antiamebic; Antiarthritic/Antirheumatic; Antimalarial; Lupus Erythematosus Suppressant